Will A Tavr To Repair An Aortic Valve Stop The Mitral Valve From Leaking?
Paravalvular prosthetic leak (PVL) or regurgitation is a serious and under-recognized condition affecting six-15% of surgical prosthetic valves and annuloplasty rings.ane,2 Moderate to astringent PVL after surgical or transcatheter aortic valve replacement (TAVR) is associated with increased mortality.3 PVL can affect any valve in whatsoever position just is more common with mitral mechanical prostheses, supra-annular aortic prostheses,1 and use of sutures without pledgets or continuous sutures in the mitral position.4 PVL can present with congestive center failure symptoms in patients with meaning degree of regurgitation. Due to relative noncompliance of the receiving sleeping accommodation, even a small volume of regurgitation can markedly increase left atrial force per unit area and induce clinical symptoms. Further elevation of pulmonary arterial pressures can lead to correct-sided heart failure. PVL can also result in significant hemolytic anemia, more commonly encountered in patients with smaller defects and high-velocity jets, patients with pre-existing anemia, and patients with increased red blood prison cell fragility due to iron or folate deficiency.v
Paravalvular defects usually consequence from poor tissue quality related to friability from infection or calcification around the prosthetic valve. Because of the underlying anatomy, repeat surgery may non exist successful in the presence of poor tissue integrity. Transcatheter PVL closure at present offers a viable and less invasive alternative in such patients. The 2022 American College of Cardiology/American Middle Clan guidelines for the management of valvular center illness recommended percutaneous repair of PVL (Class IIa) for loftier-risk surgical patients with either intractable hemolysis or New York Heart Clan (NYHA) Class Iii or Four middle failure who take anatomic features suitable for catheter-based therapy and when performed in centers with expertise in this procedure.6
Evaluation of PVL
Evaluation of PVL is challenging. Echocardiography is the master imaging modality for patients with mitral PVL, and computed tomography (CT) tin can provide condiment information (Figure i). Although two-dimensional (2D) transthoracic echocardiography (TTE) or transesophageal echocardiography (TEE) can identify the presence of PVL, three-dimensional (3D) TEE is required for perimitral leaks for complete and accurate depiction of these defects. The principal aim of imaging is to define the defect (size, circumferential extent, location, and relation to the prosthetic valve leaflets) and the state of the ventricles. It is also vital to dominion out any active endocarditis, excessive rocking move of the valve, or significant valvular regurgitation, all of which preclude percutaneous repair. Cardiac CT allows further definition of the size and orientation of PVL in cases in which acoustic shadowing affects interpretation of echocardiographic images but is non needed routinely. Cardiac magnetic resonance (CMR) with accurate flow-imaging and book-based measurements may be useful in certain cases in quantification of multiple paravalvular defects.7,8
Effigy 1
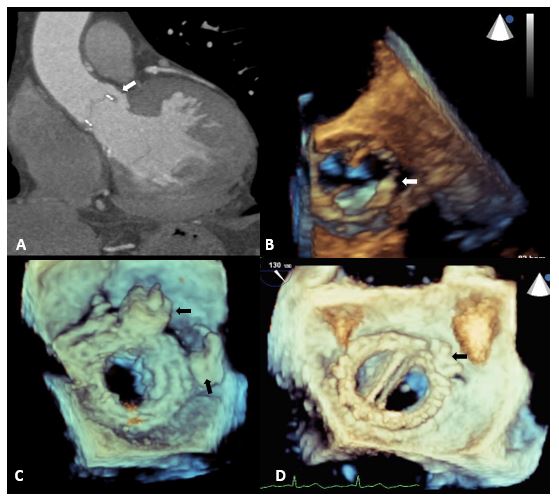
For periaortic lesions, 2D TTE (and sometimes intra-cardiac echocardiography) allows for adequate visualization for anteriorly located lesions, and TEE is better for posterior aortic defects. Aortic PVL can be difficult to quantify at times, either due to strong color menstruation Doppler signals occupying the relatively pocket-size LV outflow tract (leading to overestimation) or acoustic shadowing (resulting in underestimation). The reverberation artefact and acoustic shadowing from the posterior valve band can limit visualization of anterior PVLs even with 3D TEE. Aortography can be helpful to quantify regurgitation in such cases, although the inability to spatially characterize the leak and accurately place orthogonal angles limits its value. We rarely use this. Electrocardiography-gated cardiac CT angiography can easily define location, size, shape, and trajectory of aortic paravalvular defects. This information allows for pre-procedural determination of optimal fluoroscopy plane or gantry angles to be used during the procedure.
No affair what imaging techniques are used, careful image review and give-and-take among interventional cardiologist, echocardiographer, and cardiac radiologist is a primal pre-procedural step. Operators should understand precisely where the leak is earlier attempting to shut and what technique is mostly probable to be successful.
Transcatheter Techniques for PVL Closure
There are no specific devices that have been designed or canonical by the Food and Drug Administration (FDA) for PVL closure. PVLs vary in shape and tend to exist crescentic with long serpiginous tracks rather than cylindrical, making it difficult for a single, round device to occlude leaks. The nearly commonly used occluder devices include the AMPLATZER Vascular Plug (AVP) family (St. Jude Medical; St. Paul, MN), comprising the AVP II and AVP Four (available in the United States) and AVP III (available in Europe) (Effigy 2). Successful PVL closure requires careful pre-procedural planning and evaluation of diagnostic imaging every bit well as knowledge of prosthetic valves and their radiographic advent. Skills in circuitous catheter techniques including transseptal and (sometimes) LV apical puncture, wire snaring and creation of rails, and vascular plug delivery are necessary.5
Effigy 2: AVP Family unit

Mitral PVL Closure
Mitral PVL closure (Figure 3) is usually done under general anesthesia due to the need for intra-procedural TEE guidance. In the most commonly used "antegrade transseptal" approach, in one case the transseptal puncture is made, a 125-cm 5F multipurpose diagnostic catheter telescoped through a 6F 100-cm guide catheter (most unremarkably a multipurpose) is avant-garde into the left atrium (LA) through a steerable transseptal sheath. Use of a flexible LA sheath allows rapid access to even the near challenging medial locations adjacent to the interatrial septum. An substitution-length extra-support angled hydrophilic 0.035-inch wire passed through this telescoped catheter system is used to cantankerous the defect guided past both biplane fluoroscopy and 3D TEE. Once the wire is in a stable position beyond the defect (usually advanced into the ascending aorta), the guide catheter is avant-garde into the LV. An AVP 2 of ≤12 mm will fit through a 6F coronary guide catheter without difficulty. If a larger device is required or if multiple devices need to be placed in a nested fashion, the telescoping system is exchanged for a Flexor Shuttle Sheath (Cook Medical; Bloomington, IN), advanced beyond the defect over an Amplatz Extra-Strong wire (Melt Medical; Bloomington, IN) positioned in the LV. In the "anchor wire" technique for sequential delivery of multiple AVPs, after an AVP is deployed, the Flexor Shuttle Sheath is removed and then reloaded onto the guidewire and re-advanced across the defect, leaving the device on its delivery cablevision exterior the sheath. This sequence can exist repeated equally many times equally needed for successful closure of the defect. In example further support is needed, multiple devices can be deployed using a "stable A-Five rail" technique. The hydrophilic wire in this technique is snared in the ascending aorta and exteriorized via femoral arterial access, forming an arteriovenous loop/track. The catheters are and so removed and replaced with a Flexor Shuttle Sheath, which is avant-garde through the defect while a second operator holds tension on both ends of the exteriorized wire to provide stability. Multiple devices tin then be deployed in a fashion like to the anchor wire technique. Major advantages of the anchor wire and rail techniques include the ability to evangelize multiple devices without losing access and reversibility of the procedure. The rail technique provides the added advantage of stability and complete control of wire tension.
Figure iii: PVL Closure of a Mitral Valve-in-Valve
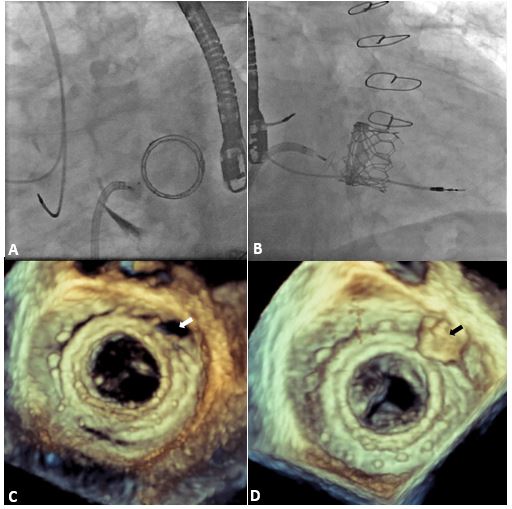
A "retrograde arroyo" involves crossing the defect from the LV into LA using a retroflexed diagnostic catheter through the aortic valve. The wire can and then exist snared in the LA and exteriorized via the transseptal arroyo forming an arteriovenous loop/track. Occluder devices tin and then be deployed in an antegrade fashion via the stable rail technique described above. "Transapical LV puncture" is ordinarily reserved for medial defects when catheter manipulation in the LA is limited or when interatrial septum is not suitable for transseptal approach (e.g., previous septal closure device). Defects are again crossed in the retrograde style equally described.
Aortic PVL Closure
This is washed most commonly in a retrograde fashion using a 125-cm 5F multipurpose diagnostic catheter telescoped through a 100-cm 6F multipurpose guide catheter. An Amplatz Left i coronary guide may be used for inductive defects, and a Judkins Right 4 catheter (Cordis; Miami Lakes, FL) might be better for some posterior defects. Later crossing the defect with a hydrophilic glidewire, the guide catheter is advanced into the LV. Occluder devices can then be deployed, or the glidewire can exist exchanged for an Amplatz Actress-Stiff wire (and the guide catheter exchanged for Flexor Shuttle Sheath) for extra stability. The ballast wire technique described above can also exist used again for sequential commitment of multiple devices. If needed for actress stability, an arterio-arterial loop/rail can too be established by snaring the hydrophilic wire (advanced through the prosthetic valve) in the ascending/descending aorta and exteriorizing it to the contra-lateral femoral artery. Transapical access with or without creating a rail might also be used in some cases as described.
PVL Closure Later on TAVR
The incidence of PVL after TAVR has declined due to improved designs (e.m., sealing skirts), CT-guided optimal valve sizing, and operator experience. However, PVLs can still occur due to valve malposition, under expansion, or heavy annular/LV outflow tract calcification. The first two are treated by a valve-in-valve or balloon post-dilation, respectively, and PVL closure using a vascular plug is the treatment modality of option for the last. Retrograde wire cannulation is occasionally challenging due to native valves-in-situ, bulky calcification, and the presence of sealing skirts in newer devices. The aortic frame struts present boosted challenge in case of the CoreValve (Medtronic; Dublin, Republic of ireland) for crossing the defect while avoiding the lattice. A high crossing position across the frame struts allows relatively easier deliverability of vascular plugs. AVP 4 are commonly used for PVLs associated with TAVR due to smaller size and heavy calcification encountered in such defects. AVP Iv range in size iv-8 mm and are easier to deliver through a 0.038-inch lumen catheter (including a 4F multipurpose catheter). This reduces the demand to exchange for larger catheters/sheaths and avoids excessive manipulation.
Outcomes
Successful PVL closure is divers every bit reduction of regurgitation to grade ≤ane, is achieved in ~70-90% of cases, and improves with increasing operator experience.9,ten Currently, 90% of fifty-fifty the most circuitous leaks can be closed successfully and without complications.10,11 Previous studies take demonstrated that a successful procedure with reduction of residual regurgitation to the lowest possible grades translated into superior clinical outcomes. This was seen in a previous meta-analysis (n = 362) in which successful PVL closure (compared with a failed intervention) was associated with significant improvement in NYHA class or hemolytic anemia (odds ratio, 9.95; 95% confidence interval, 2.1-66.seven), reduction in demand for cardiac surgery (odds ratio, 0.08; 95% confidence interval, 0.01-0.40), and, well-nigh chiefly, lower cardiac bloodshed (odds ratio, 0.08; 95% confidence interval, 0.01-0.xc).12
In a recently published analysis of 86 aortic PVL closures, Alkhouli et al. demonstrated successful reduction of residue regurgitation to mild or less in 62% of the cases with major adverse cardiac issue rate of v.1% at thirty days.13 In another previously published serial of aortic and mitral PVL closures from Mayo Dispensary, the 30-day complexity rate was 8.7% (sudden and unexplained death was 1.7%; stroke was 2.six%; emergency surgery was 0.9%; and haemorrhage was v.2%).11 Importantly, a significant comeback in procedural outcomes was noted with increasing operator experience in this study.11 This highlights the complication and steep learning curve associated with this process, which requires commitment and a wide variety of procedural skills.
Important procedural complications can include prosthetic leaflet impingement (4%) and device embolization (<1%).five Prosthetic leaflet impingement is more common in mechanical and stentless prosthesis, larger occluder devices with overhang. If it occurs, the occluder device needs to be readjusted or switched out for a smaller device. The anterior-superior defects are likewise prone to device movement once released. Device embolization tin can exist prevented in most cases past ensuring stability of the device past pushing on it using the sheath/catheter while pulling on the delivery cablevision before releasing it. The device can be repositioned or changed for a larger device in case of instability. If the device embolizes, it usually volition club in a common iliac avenue, although smaller devices tin embolize cranially. Embolized devices can be snared into the arterial sheath and removed. Aortography or selective coronary angiography might exist necessary to rule out coronary obstruction that can occur with aortic PVL closure. Transapical access tin be associated with postoperative bleeding (~20%) and hemothorax, requiring chest tube drainage especially when 6F sheath is used. Of notation, apical puncture sites can exist airtight with vascular plugs when larger sheath sizes are used.
Future Directions
The outcomes of PVL closure have significantly improved over the years with growing experience and pre-procedural planning using advanced imaging modalities. There are little data on invasive hemodynamics that might provide procedural guidance besides as prognostic information about long-term outcomes following PVL closure. Most importantly, devices specifically designed for this purpose would likely amend outcomes and are in development. Advancements in intra-procedural imaging guidance similar the use of fusion imaging with CT overlay during fluoroscopy will besides be useful in facilitating the procedure.14
Conclusion
Successful PVL closure can be performed with low complication rates if washed by experienced operators at centers of expertise. Conscientious pre-procedural planning, intra-procedural imaging, and experience in circuitous catheter skills are necessary to optimize procedural success. Every attempt should exist made to achieve the lowest possible remainder regurgitation to ensure superior clinical outcomes.
References
- Ionescu A, Fraser AG, Butchart EG. Prevalence and clinical significance of incidental paraprosthetic valvar regurgitation: a prospective written report using transoesophageal echocardiography. Heart 2003;89:1316-21.
- O'Rourke DJ, Palac RT, Malenka DJ, Marrin CA, Arbuckle Exist, Plehn JF. Result of mild periprosthetic regurgitation detected by intraoperative transesophageal echocardiography. J Am Coll Cardiol 2001;38:163-6.
- Sponga Due south, Perron J, Dagenais F, et al. Impact of residual regurgitation after aortic valve replacement. Eur J Cardiothorac Surg 2022;42:486-92.
- Englberger L, Schaff HV, Jamieson WR, et al. Importance of implant technique on risk of major paravalvular leak (PVL) subsequently St. Jude mechanical heart valve replacement: a study from the Artificial Valve Endocarditis Reduction Trial (Avoid). Eur J Cardiothorac Surg 2005;28:838-43.
- Eleid MF, Cabalka AK, Malouf JF, Sanon S, Hagler DJ, Rihal CS. Techniques and Outcomes for the Handling of Paravalvular Leak. Circ Cardiovasc Interv 2022;eight:e001945.
- Nishimura RA, Otto CM, Bonow RO, et al. 2022 AHA/ACC Guideline for the Management of Patients With Valvular Centre Affliction: executive summary: a study of the American Higher of Cardiology/American Heart Association Task Forcefulness on Practice Guidelines. Circulation 2022;129:2440-92.
- Lázaro C, Hinojar R, Zamorano JL. Cardiac imaging in prosthetic paravalvular leaks. Cardiovasc Diagn Ther 2022;4:307-13.
- Pflaumer A, Schwaiger M, Hess J, Lange R, Stern H. Quantification of periprosthetic valve leakage with multiple regurgitation jets past magnetic resonance imaging. Pediatr Cardiol 2005;26:593-4.
- Sorajja P, Cabalka AK, Hagler DJ, Rihal CS. The learning curve in percutaneous repair of paravalvular prosthetic regurgitation: an analysis of 200 cases. JACC Cardiovasc Interv 2022;7:521-9.
- Ruiz CE, Jelnin V, Kronzon I, et al. Clinical outcomes in patients undergoing percutaneous closure of periprosthetic paravalvular leaks. J Am Coll Cardiol 2022;58:2210-7.
- Sorajja P, Cabalka AK, Hagler DJ, Rihal CS. Percutaneous repair of paravalvular prosthetic regurgitation: acute and 30-day outcomes in 115 patients. Circ Cardiovasc Interv 2022;four:314-21.
- Millán X, Skaf Southward, Joseph L, et al. Transcatheter reduction of paravalvular leaks: a systematic review and meta-assay. Can J Cardiol 2015;31:260-9.
- Alkhouli M, Sarraf Thou, Maor East, et al. Techniques and Outcomes of Percutaneous Aortic Paravalvular Leak Closure. JACC Cardiovasc Interv 2022;9:2416-26.
- Cruz-Gonzalez I, Rama-Merchan JC, Rodríguez-Collado J, et al. Transcatheter closure of paravalvular leaks: state of the art. Neth Heart J 2022;25:116-24.
Keywords: Anemia, Hemolytic, Aorta, Thoracic, Aortic Valve, Aortography, Arterial Pressure, Atrial Pressure, Angiography, Coronary Angiography, Echocardiography, Iii-Dimensional, Echocardiography, Echocardiography, Transesophageal, Electrocardiography, Endocarditis, Femoral Artery, Fluoroscopy, Folic Acrid, Heart Atria, Heart Failure, Middle Valve Diseases, Hemothorax, Iliac Avenue, Magnetic Resonance Spectroscopy, Mitral Valve, Stroke, Transcatheter Aortic Valve Replacement
< Back to Listings
Will A Tavr To Repair An Aortic Valve Stop The Mitral Valve From Leaking?,
Source: https://www.acc.org/latest-in-cardiology/articles/2017/02/02/08/25/transcatheter-management-of-paravalvular-leaks
Posted by: carverdoely1938.blogspot.com


0 Response to "Will A Tavr To Repair An Aortic Valve Stop The Mitral Valve From Leaking?"
Post a Comment